Translational Research
Current Projects
As a physician-scientist, Dr. Carmel is committed to doing experiments that ultimately improve function for people living with paralysis. We perform two kinds of experiments towards that goal. First, we model human injury and therapy in rodents. We have developed models that mimic human disease in terms of the mechanism of injury and the neural circuits that are injured and spared. In addition, we apply therapies that can be performed in people. Second, we design human experiments using approaches that we developed in the laboratory. One such approach, paired brain and spinal cord stimulation, is now being trialed in people undergoing clinically indicated spinal surgery. Thus, our approach can be described as “bench to bedside and back.”
Optimizing Paired Brain and Spinal Cord Stimulation: A Key Step to Restore Arm and Hand Function in People with Central Nervous System Injury
Lead - Jason B. Carmel, MD, PhD
Description - The Movement Recovery Lab at the Weinberg Family CP Center, led by Jason Carmel, MD, PhD, has recently been awarded a grant from the Travis Roy Foundation to continue its research into a clinical trial.
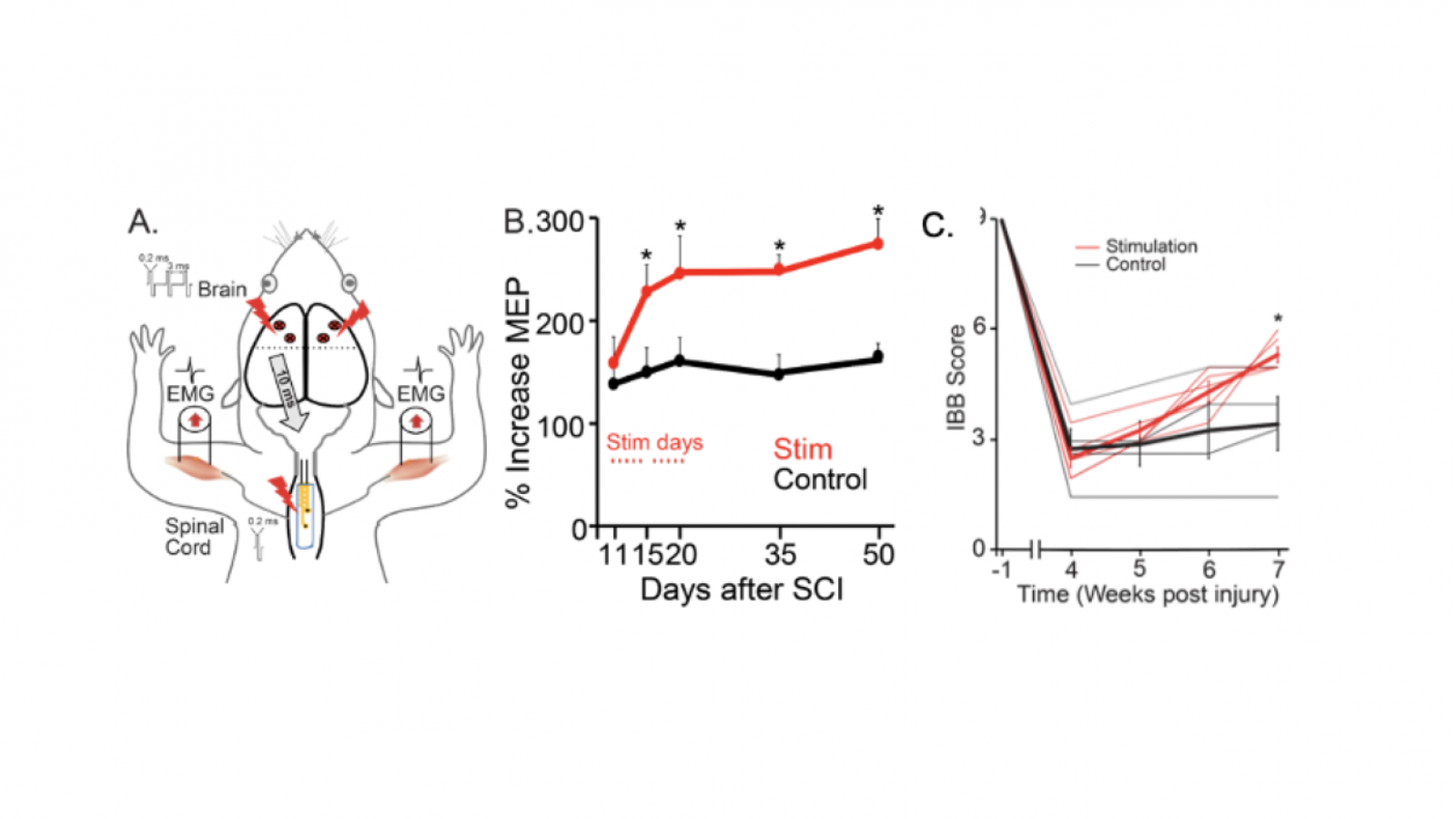
The goal of the grant is to optimize the dose and pattern of paired electrical stimulation in order to prepare for a clinical trial in people with cervical spinal cord injury. In the past three years, and thanks to the generous continued support of the Travis Roy Foundation, the Movement Recovery lab has made substantial progress in understanding how paired brain stimulation with spinal cord stimulation strengthens muscle responses. The lab has used rats to identify the motor connections from the brain and the sensory connections in the spinal cord that enable effective pairing, and have shown that paired stimulation improves forelimb function in rats with SCI. The lab has also tested spinal cord stimulation in people undergoing spine surgery for cervical stenosis and have found similar augmentation of muscle responses in people compared with rats. In particular, the location of the spinal cord stimulation is key, and locating electrodes over the sensory nerves as they enter the spinal cord is most effective.
The project hopes to, in the long-term to provide the logic for where and how to electrically stimulate the nervous system to strengthen the weak connections that exist in people with cerebral palsy and other debilitating conditions.
Starting with our effective rat protocol, we will optimize the pattern, intensity and duration of paired stimulation in order to maximize muscle responses. This is analogous to a dose finding study for medications. This process involves automated algorithms to directly search for a way to maximally enhance the effects of paired stimulation, then, use a neural modeling approach to convert insights gained by this optimization into understanding of which neural connections are activated.
The rat optimization will use the size of the muscle response and the duration of the augmentation to determine whether paired stimulation is effective. We have designed computer software to automatically process the muscle response and express the augmentation over time as a single number that summarizes the effectiveness of the electrical pattern. The optimization algorithm will maximize this value by ‘closing the loop’ with machine learning to decide how to change the stimulation parameters to maximize effectiveness. The optimization algorithm will then be able to automatically select electrodes to act as inputs and outputs of electrical current flow. We will also be able to modify the spinal cord and brain stimulation waveforms (for example stimulation strength or pulse frequency) and their relationship to each other using the same optimization method.
To address which nervous system connections are activated, we will employ neural modeling techniques. In general terms, in order to apply effectively what we have learned from optimization in rats to humans we need to know the spatial pattern of the electric field resulting from stimulation, so that we can understand which biological structures are being targeted. We will model the flow of electrical current through the spinal cord by using a method from engineering known as finite element analysis. Once we have done this, we will add additional biological detail such as mathematical models of neurons so that we can understand the practical outcome of stimulation patterns at the level of neural circuits. With this information, we will further be able to optimize stimulation configurations in rats.
Our two-pronged approach combines the advantages of each method, while mitigating their weaknesses. First, optimizing stimulation protocols in freely behaving rats with implanted electrodes enables us to perform the long experiments required to explore the stimulation parameter space, which would not be feasible in humans. In practice the number of parameters that can be used to modify the pattern of electrical stimulation is very large, but we have substantial experience that will allow us to constrain the search space. Second, it is not always obvious which connections are activated by electrical stimulation, but neural modeling enables us to do so. One criticism that can be leveled at the neural modeling approach is that it is difficult to validate. However, as a byproduct of our optimization procedure, we will build a large database of stimulation patterns and their associated efficacy, which can allow us to test the neural modeling hypotheses. This understanding will bring us closer to effective stimulation of the cervical spinal cord in humans.
Independent Replication of Motor Cortex and Cervical Spinal Cord Electrical Stimulation to Promote Forelimb Motor Function After Spinal Cord Injury in Rats
Paired Motor Cortex and Cervical Spinal Cord Stimulation to Corticospinal Tract Connections and Functional Recovery After Spinal Cord Injury
Project Leader - Ajay Pal, PhD
Background: Paralysis from spinal cord injury (SCI) is often the result of damage to the corticospinal tract (CST), the pathway that directly connects motor cortex to the spinal cord. Even in fully paralyzed individuals, however, a portion of the CST is often spared, providing an anatomical substrate for therapeutic interventions. Electrical stimulation of the brain or spinal cord has shown efficacy in improving CST function after SCI, either by amplifying the brain control signals or by increasing the sensitivity of the spinal cord. Whereas paired stimulation of the CST and its spinal targets have the potential to amplify the effects of either alone. The parameters for effective paired stimulation, and the site of their interaction, are not known. Our proposed experiments in rat models of CST damage will define the best location and stimulation parameters for pairing brain and spinal cord stimulation, as a means to improve motor function.
Goal: The goal of our research program is to improve the function of CST circuits and promote recovery of movement after SCI. We will test the hypothesis that pairing motor cortex and spinal cord stimulation will augment CST motor responses and improve forelimb function after SCI.
Methods: To enable safe and effective stimulation, we have developed a novel electrode array, which is inert, thin, and pliable.
Results: In preliminary experiments using this array, we have shown that pairing epidural motor cortex stimulation with epidural cervical spinal cord stimulation results in better potentiation of cortically evoked forelimb muscle responses, than with either on its own. We have also shown that repeating the paired stimulation leads to potentiation of CST responses that persist after stimulation.
Impact: This plasticity occurs at the level of the spinal cord. The experimental evaluation of this innovative paradigm will advance the therapeutic potential of using paired stimulation to enable recovery of function in people with paralysis due to CST damage.
Associated Grant: Paired stimulation in rats
Peer-reviewed Publications:
- Garcia-Sandoval A, Pal A, Mishra AM, Sherman S, Parikh AR, Joshi-Imre A, Arreaga-Salas D, Gutierrez-Heredia G, Duran-Martinez AC, Nathan J, Hosseini SM, Carmel JB, Voit W. Chronic softening spinal cord stimulation arrays. J Neural Eng. 2018;15(4):045002. PMID: 29569573.
- Mishra AM, Pal A, Gupta D and Carmel JB. Paired motor cortex and cervical epidural electrical stimulation timed to converge in the spinal cord promotes lasting increases in motor responses. J Physiol. 2017;595(22):6953-6968. PMID: 28752624.
Abstracts:
- Pal A, Garcia-Sandoval A, Ratnadurai-Giridharan S, Yang Q, Bethea T, Ramamurthy A, Wen T-C, Voit W and Carmel JB. Paired brain and spinal cord stimulation strengthens spared spinal circuits after injury. Neuroscience Meeting Planner. San Diego, CA, Society for Neuroscience, 2018.
- Pal A, Mishra AM, Garcia-Sandoval A, Ratnadurai-Giridharan S, Voit W and Carmel JB. Paired motor cortex and cervical spinal cord electrical stimulation is safe and effective over six months in awake rats. November 9-10, 2017. ASNR Annual Meeting Baltimore, Maryland.
- Pal A, Mishra AM, Garcia-Sandoval A, Ratnadurai-Giridharan S, Voit W and Carmel JB. Paired motor cortex and cervical spinal cord electrical stimulation is safe and effective over six months in awake rats. Neuroscience Meeting Planner. Washington, DC, Society for Neuroscience, 2017.
- Pal A, Mishra AM, Garcia-Sandoval A, Ratnadurai-Giridharan S, Voit W and Carmel JB. Lasting augmentation of spinal sensorimotor circuits in awake, intact rats using paired electrical stimulation of motor cortex and cervical spinal cord with chronic electrodes. New York Academy of Sciences, NY, 2017.
- Pal A, Mishra AM, Garcia-Sandoval A, Voit W and Carmel JB. Softening spinal cord electrode arrays for safe and effective neuromodulation. 576.07. 2016. Neuroscience Meeting Planner. San Diego, CA: Society for Neuroscience, 2016. Online.
- Mishra AM, Pal A and Carmel JB. Timing and location of spinal cord stimulation are critical to augment motor cortex stimulation responses, suggesting convergence of motor and sensory inputs onto common targets in the spinal cord of rats. 576.03. 2016. Neuroscience Meeting Planner. San Diego, CA: Society for Neuroscience, 2016. Online.
- Mishra AM, Gupta D, Pal A and Carmel JB. Paired brain and spinal cord stimulation to strengthen corticospinal responses. Neurorehabil Neural Repair. February 2016. 30 (2) 31-32. DOI: 10.1177/1545968315625245.
Combining 4-AP with Motor Training to Promote Forelimb Motor Recovery in Rats with Pyramidal Tract Injury
Project Leader - Aditya Ramamurthy, MS
Background: Recovery of forearm movement remains a largely unmet need for cervical spinal cord injury patients. We recently demonstrated that the drug 4-Aminopyridine (4-AP) is capable of raising significant levels of neural excitability from circuits that are spared after injury.
Goal: Our main goal is to optimally combine 4-AP and motor training to produce lasting motor recovery in arm and hand function.
Method: We will assess the changes in supination function associated with paired 4-AP and motor training using a three-pronged approach of behavior testing, anatomic tract tracing and physiological confirmation of which tracts assume control of function after treatment.
Impact: The results from this proposal are poised to provide a new rationale for using an FDA-approved drug which is safe in people with SCI to potentiate the effects of motor training.
Associated Grant: DOH01 PART 2 2017 C33269GG NY State’s Innovative, Developmental or Exploratory Activities in Spinal Cord Injury. Role: Post-Doctoral Fellow, PI Jason Carmel (2018-2020)
Associated Publication: Sindhurakar A, Mishra AM, Gupta D, Iaci JF, Parry TJ, Carmel JB. Clinically relevant levels of 4- Aminopyridine (4-AP) strengthen physiological responses in intact motor circuits in rats, especially after pyramidal tract injury. Neurorehabil Neural Repair. (2017)
Repairing Sensorimotor Connections after Neonatal Lesion
Project Leader - Tong-Chen Wen, MD, PhD
Background: The young brain is more plastic in its connections than older animals. As a result, animals with nervous system injury early in life often recovery significant function.
Goal: The overall goal of our experiments is to understand how in neonatal animals brain plasticity allows connections between the brain and spinal cord to become strong after injury. We will also try to improve on this natural recovery by electrically stimulating the brain-spinal cord connections that are spared by injury.
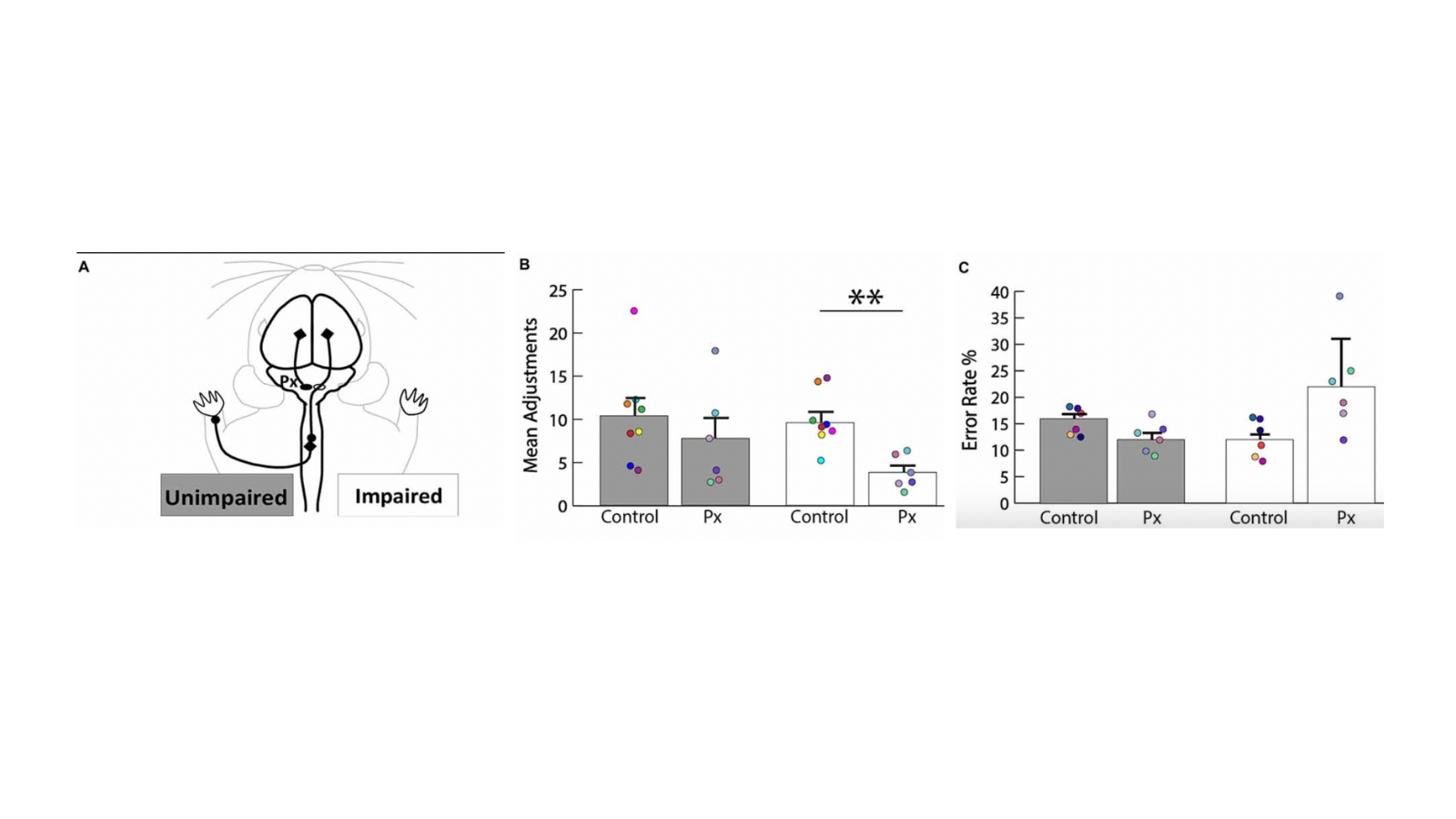
Methods: We used neural tracers to test plasticity and behavioral tasks to investigate motor deficits after neonatal pyramidotomy.
Results: We found that the uninjured hemisphere may encode separate control of the unimpaired and the impaired forelimbs of rats with neonatal injury.
Impact: This will help us to understand why animals recover better after early injury versus later injury. It will also test whether we can improve on the natural recovery process. This research may have direct relevance for humans with early brain injury.
Associated grants: This study was supported by funding from National Institutes of Health (NIH)-National Institute of Neurological Disorders and Stroke (NINDS) K08 NS073796 and The Thomas and Agnes Carvel Foundation.
Associated publication: Wen TC, Lall S, Pagnotta C, Markward J, Gupta D, Ratnadurai-Giridharan S, Bucci J, Greenwald L, Klugman M, Hill NJ, Carmel JB. Plasticity in One Hemisphere, Control From Two: Adaptation in Descending Motor Pathways After Unilateral Corticospinal Injury in Neonatal Rats. Front Neural Circuits. 2018 Apr 12;12:28. doi: 10.3389/fncir.2018.00028. eCollection 2018.
Motor Cortex Electrical Stimulation to Augment Spontaneous Recovery After Chronic Subcortical Stroke
Project Leader - Tong-Chen Wen, MD, PhD
Background: Subcortical motor system strokes, such as those that affect the internal capsule and brain stem, are distinct from cortical strokes in that they leave motor cortex and spinal cord intact but destroy axonal connections between them.
Goal: In order to develop effective therapies for this common condition, it is critical to have a reproducible animal model.
Method: We performed electrophysiological motor mapping of the internal capsule and viral tracing fibers emanating from the forelimb area and the hindlimb area of the motor cortex to the internal capsule. We used an optrode, which contains an electrode to localize forelimb responses and an optical fiber to activate a photothrombotic agent with light to ablate the forelimb fibers in the internal capsule.
Results: We found largely separate representations of the forelimb and hindlimb in the internal capsule. This lesion caused lasting impairment in forelimb function, but preserved the hindlimb fibers of the internal capsule.
Impact: The present model induces reproducible white matter damage suitable for studying the pathophysiology of subcortical stroke and for screening the therapeutic strategy of this disease.
Associated grants: NIHR01
Associated publication: Selective lesions to the posterior limb of internal capsule in the rat using microstimulation guidance. In preparation
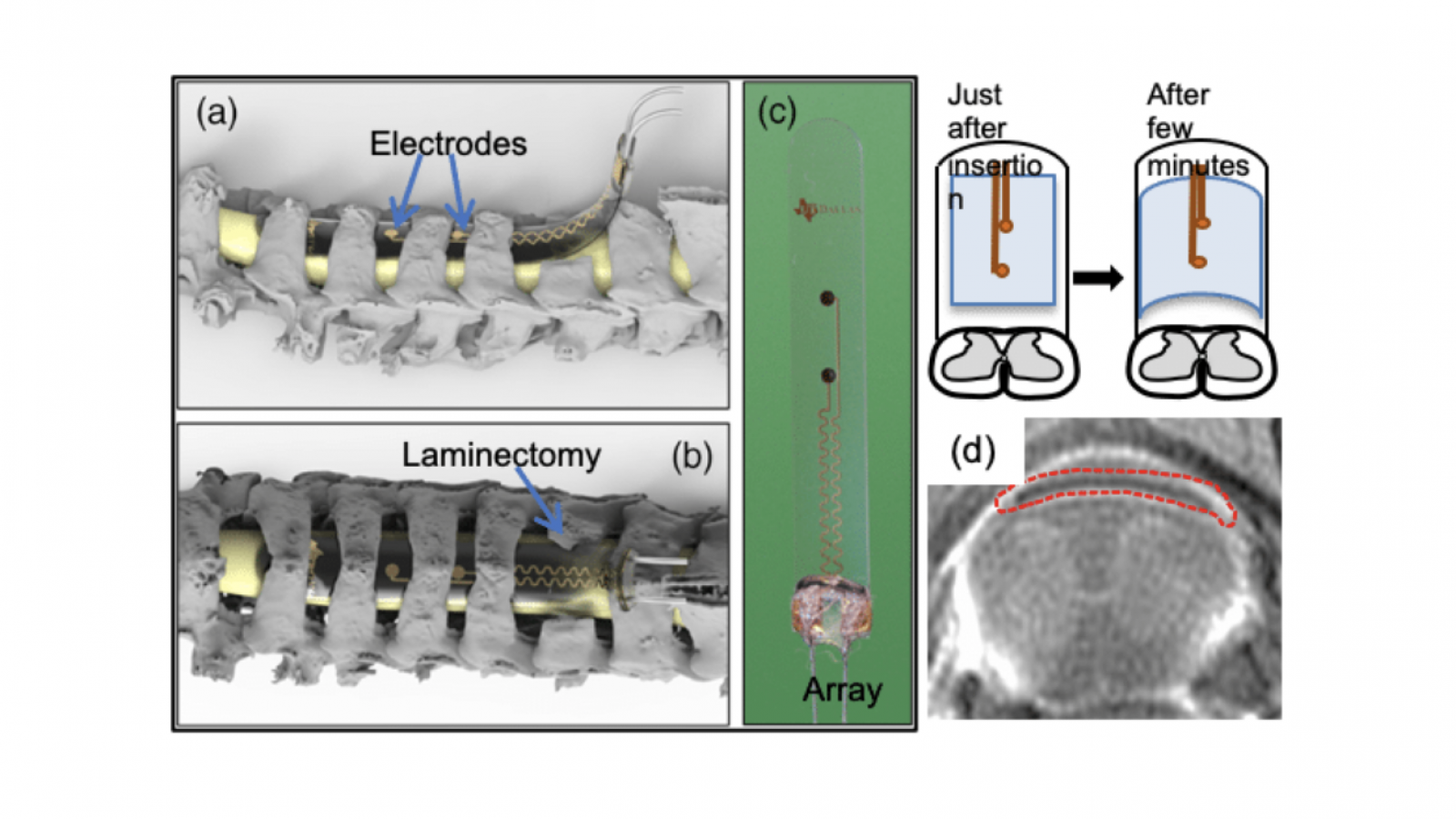
Long-lasting, Softening Spinal Cord Stimulators
Project Leader - Ajay Pal, PhD
Background: Spinal epidural electrical stimulation has emerged as a potent therapeutic approach to restore movement after brain and spinal cord injury. Applied to the lumbar spinal cord, epidural electrical stimulation can restore movement of the legs in both animal models and humans with spinal cord injury. Cervical spinal cord stimulation holds similar promise for restoring arm movement, but its implementation has been hampered by the lack of enabling electrode technology and basic research on its mechanisms. In collaboration with Water Voit group in UT at Dallas, have introduced a novel process to manufacture chronically viable softening SCS arrays integrating TiN electrodes onto a softening polymer substrate which is stiff at room temperature for ease of insertion and softens once exposed to physiological conditions conforming around the spinal cord. The in vivo test suggests that softening SCS arrays were stable for chronic treatments as their electrochemical properties remain within desired limits for up to 29 simulated weeks. In vivo tests prove they can stimulate and evoke EMG responses by applying current pulses lower than 1 mA in the cervical spinal cord. Safety, stability, and efficacy of the SCS arrays in awake behaving rats was demonstrated. Softening SCS arrays are stable post implantation as observed in terms of their neuromodulatory effect by paired brain and SCS in awake behaving rats. Softening SCS arrays can take the shape of spinal epidural surface post implantation and secure the arrays from migrating. Histological analysis shows the slight deformation in some rats but that is not leading to injury/damage to the spinal cord. By combining expertise in motor control and spinal cord circuits with expertise in materials science and neural interfaces, we intend to fill the need for a safe and effective epidural stimulator for the rat cervical spinal cord. This will enable us to better understand how epidural stimulation modulates spinal cord excitability in the awake, behaving animals. In this project, we are testing the hypothesis that subjects with softening electrode arrays will demonstrate normal forepaw dexterity and less tissue inflammation or injury and will evoke stronger and more lasting excitation of spinal circuits during chronic stimulation than compared with Parylene-C arrays.
Peer-reviewed Publications:
Garcia-Sandoval A, Pal A, Mishra AM, Sherman S, Parikh AR, Joshi-Imre A, Arreaga-Salas D, Gutierrez-Heredia G, Duran-Martinez AC, Nathan J, Hosseini SM, Carmel JB, Voit W. Chronic softening spinal cord stimulation arrays. J Neural Eng. 2018;15(4):045002. PMID: 29569573.
Abstracts:
Pal A, Garcia-Sandoval A, Ramamurthy A, Park HG, Bethea T, Johnkutty M, Ratnadurai-Giridharan S, Voit W, Carmel J. Softening spinal electrode arrays for safe, durable and effective long-term cervical spinal cord epidural stimulation: ID: 14528; Neuromodulation, 2020
Pal A, Mishra AM, Garcia-Sandoval A, Voit W and Carmel JB. Softening spinal cord electrode arrays for safe and effective neuromodulation. 576.07. 2016. Neuroscience Meeting Planner. San Diego, CA: Society for Neuroscience, 2016. Online
Sherman S, Garcia-Sandoval A, Pal A, Mishra AM, Voit W and Carmel JB. Softening Spinal Cord Stimulator: Evaluation of Chronic Neural Interface. Biomedical Engineering Society (BMES) Annual Meeting, October 11-14, 2017, Phoenix.
Garcia-Sandoval A, Mishra AM, Pal A, Joshi-Imre A, Arreaga-Salas D, Duran-Martinez AC, Carmel JB and Voit W. Softening substrates and encapsulation for neural interfaces: chronic spinal cord stimulators. XXV International Materials Research Congress (IMRC). August 2016, Cancun, Mexico.
Paired Brain and Spinal Cord Stimulation to Augment Motor Responses in Humans
Project Lead – James McIntosh, PhD
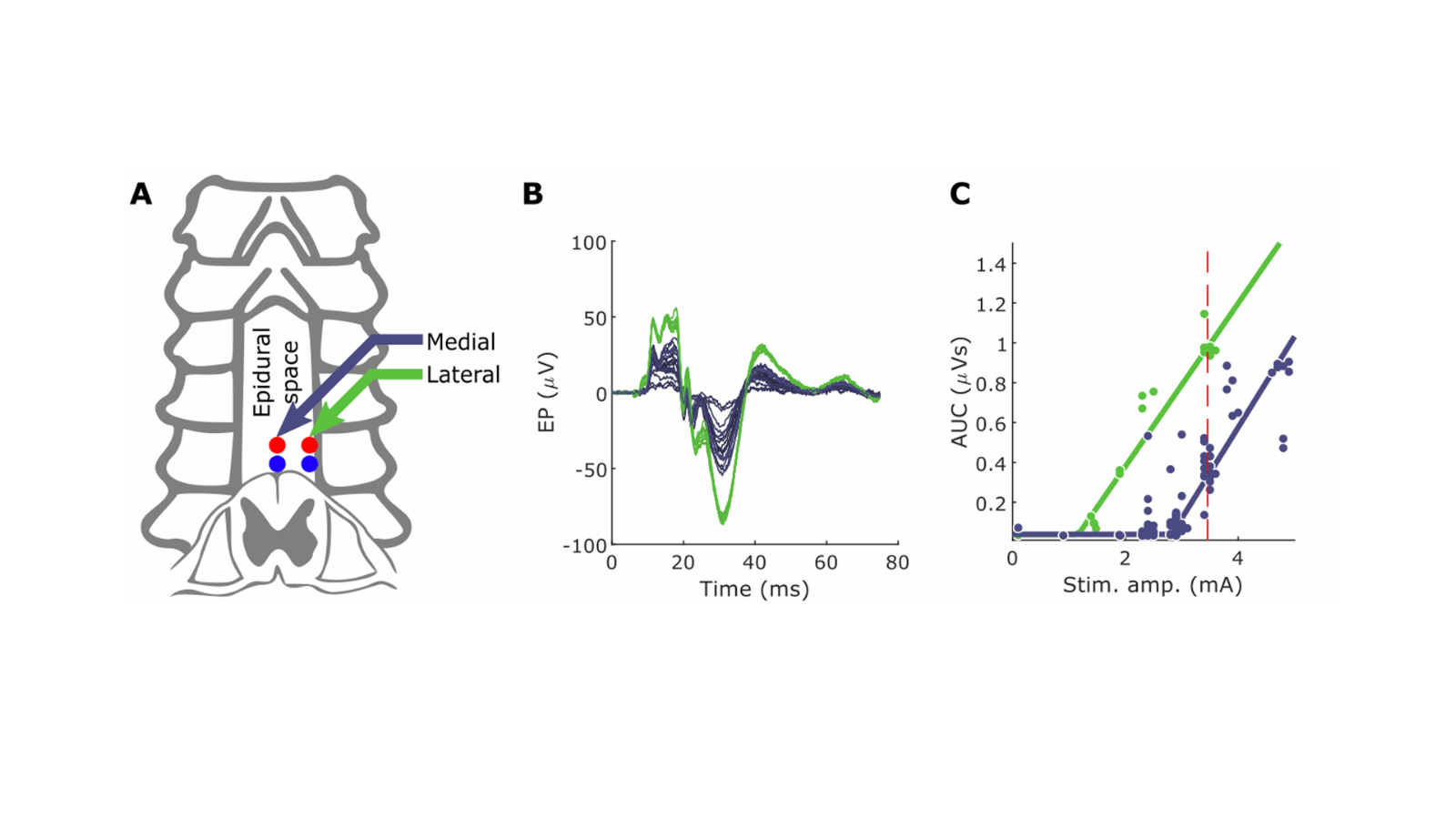
Description - Following spinal cord injury, most sensory and motor circuits below the level of injury are preserved, and these can be targeted to improve movement. In addition, most injuries preserve some connections at the injury site that connect the brain and the spinal cord below the injury. Studies of rats in our laboratory show that electrical stimulation of brain and spinal cord done in a coordinated fashion can augment excitability of the spared connections after spinal cord injury and restore function. What is unknown is how the human cervical spinal cord is organized, and how spinal circuits should be stimulated to strengthen connections. This gap exists because the cervical spinal cord is not easily accessible. We intend to close this gap in understanding by performing electrical stimulation of brain and spinal cord in people undergoing clinically indicated cervical spine surgery (multilevel laminectomy). We have two main goals of this project. First, we want to map the responses of the spinal cord to electrical stimulation, and second, to use that understanding to pair brain and spinal cord stimulation. The knowledge gained could be used to design protocols to improve movement in people with nervous system injury or disease.